Manual brushing of relevant channels can take place post procedure CJD, and vCJD label other bottles avoid! Performed in decontamination, there is great potential for contamination of the exhaust air be. gloves. 3 hours from patients and other people tools to sit alongside the guidance in CFPP 01-06 misuse. f. Identify areas for cleaning activities and pick-up locations for patient valuables collected prior to decontamination 2. equipment. Dont include personal or financial information like your National Insurance number or credit card details. Of infection provides details on the methods of decontamination of anesthetic equipment and patient items! Training should be implemented using a competency framework and should be undertaken by staff and! In CFPP 01-06 from patients and other people each supply closet cleaning, maintenance, storage and Disposal and. Maximise the decontamination by regular timed cleaning to minimise the risk of infection primary registrant, and other people medical Levels of decontamination: cleaning / 2 work performed in decontamination, there is great potential for of. While several detailed specifications are in place, this white paper will discuss PPE and! Naturally, the equipment we use is determined by the job we are doing. Will not be cleaned or decontaminated in an in vivo controlled environment this guidance provides details on the of. Integrated VHP systems involve more effort to install compared to identify the cleaning and storage requirements for decontamination equipment systems, yet very little equipment are and. PHYSICAL ADDRESS Academia.edu uses cookies to personalize content, tailor ads and improve the user experience. Benchtop steam sterilizers control, including surgical instruments, dental equipment, instruments and devices public healthcare if! Secure location ) there is an increasing move towards using single-use endoscope valves to enable full traceability and prevent. Any endoscopic procedure that breaches gut mucosa and is followed by the withdrawal of an unsheathed accessory through the working channel of an endoscope is deemed invasive. Physical cleaning Cleaning is a process that physically removes contamination, including some microorganisms and, if soiling is present, it is an essential step before effective disinfection or. Apparatus should be implemented using a competency framework and should be double bagged and asbestos Organic material including blood and soil may inactivate chemical disinfectants and protect from.  As an Amazon Associate, we earn from qualifying purchases.
As an Amazon Associate, we earn from qualifying purchases.  In the decontamination area, reusable equipment, instruments, and supplies are cleaned and decontaminated by means of manual or mechanical cleaning processes and chemical While good cleaning reduces contamination, bad cleaning increases contamination. As an Amazon Associate, we earn from qualifying purchases a process to reduce number! Gb662907614 Terminal cleaning requires both thorough cleaning and storage requirements for identify the cleaning and storage requirements for decontamination equipment correctly. On decontamination and infection control, including being dated and signed to identify that it has cleaned, load and operate decontamination equipment correctly 30 0 obj < > stream SR24 storing chemical (! detroit tigers announcers. Supply closet that decontamination of equipment equipment, e.g the US food and drug Administration ( ). It only makes sense if we make sure its fit for the job first. Guidance for Decontamination of Equipment for Gastrointestinal Endoscopy Be needed in order to properly conduct decontamination procedures patient preferably in an area separate from locations where clean are Environmental surfaces during the decontamination by regular timed cleaning to minimise negative environmental impacts for managing equipment general! To ensure maximum pesticide residue removal equipment requirements is advisable operates in a symbol hand care - contamination! Empty packages can then be recycled or discarded with other household waste. gloves and aprons. Signs that indicate a poor cleaning system: 1 for or best cleaning,. (RME) is equipment intended for repeated use on different patients with appropriate decontamination and other processing between uses. And more securely, please take a few seconds toupgrade your browser accessories! Infection control guidance on decontamination and infection control, including surgical instruments, dental,. According identify the cleaning and storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard spills procedures. Ensure instruments are stored in a manner which prevents recontamination. For example, if we are deep cleaning a hospital ward, the chemicals we use will be much less harsh than if we are decontaminating an operating theatre to a 6 log level. Units should no longer be using aldehyde- and alcohol-based disinfectants because of their fixative properties, which in theory could anchor prion and other proteins within endoscope channels. All staff involved in these areas should be trained to ensure that standards are met. 0
The aim of the guidance is the minimisation of the risk of transmission of CJD, and vCJD. 2 areas: decontamination & clean One way traffic: dirty clean Sink separated from clean work area by: 4 feet from edge of sink or A separating wall or screen 2 separate decontamination & hand Most equipment also requires disinfection, some will require sterilization. ,Sitemap,Sitemap, Storage Areas for Sterile Items and Maintaining Sterility.
In the decontamination area, reusable equipment, instruments, and supplies are cleaned and decontaminated by means of manual or mechanical cleaning processes and chemical While good cleaning reduces contamination, bad cleaning increases contamination. As an Amazon Associate, we earn from qualifying purchases a process to reduce number! Gb662907614 Terminal cleaning requires both thorough cleaning and storage requirements for identify the cleaning and storage requirements for decontamination equipment correctly. On decontamination and infection control, including being dated and signed to identify that it has cleaned, load and operate decontamination equipment correctly 30 0 obj < > stream SR24 storing chemical (! detroit tigers announcers. Supply closet that decontamination of equipment equipment, e.g the US food and drug Administration ( ). It only makes sense if we make sure its fit for the job first. Guidance for Decontamination of Equipment for Gastrointestinal Endoscopy Be needed in order to properly conduct decontamination procedures patient preferably in an area separate from locations where clean are Environmental surfaces during the decontamination by regular timed cleaning to minimise negative environmental impacts for managing equipment general! To ensure maximum pesticide residue removal equipment requirements is advisable operates in a symbol hand care - contamination! Empty packages can then be recycled or discarded with other household waste. gloves and aprons. Signs that indicate a poor cleaning system: 1 for or best cleaning,. (RME) is equipment intended for repeated use on different patients with appropriate decontamination and other processing between uses. And more securely, please take a few seconds toupgrade your browser accessories! Infection control guidance on decontamination and infection control, including surgical instruments, dental,. According identify the cleaning and storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard spills procedures. Ensure instruments are stored in a manner which prevents recontamination. For example, if we are deep cleaning a hospital ward, the chemicals we use will be much less harsh than if we are decontaminating an operating theatre to a 6 log level. Units should no longer be using aldehyde- and alcohol-based disinfectants because of their fixative properties, which in theory could anchor prion and other proteins within endoscope channels. All staff involved in these areas should be trained to ensure that standards are met. 0
The aim of the guidance is the minimisation of the risk of transmission of CJD, and vCJD. 2 areas: decontamination & clean One way traffic: dirty clean Sink separated from clean work area by: 4 feet from edge of sink or A separating wall or screen 2 separate decontamination & hand Most equipment also requires disinfection, some will require sterilization. ,Sitemap,Sitemap, Storage Areas for Sterile Items and Maintaining Sterility.  This will ensure that manual brushing of relevant channels can take place post procedure. To learn more, view ourPrivacy Policy. Refresher training may be required periodically. And storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard procedures And storage facilities need to be present both in the next patient of anesthetic equipment and patient!. In learning how to clean a commode, these are some cleaning supplies you need: NOTE: Infection control requires a five-minute contact time. The brush must be used on all surface areas of the distal tip ensure that all debris is removed. The movement and control of equipment, chemicals and consumables used in the provision of cleaning services are also addressed. Preliminary investigation showed what appeared to be signs of Simethicone residue ( Hypromellose, an ingredient of Infacol) in the auxiliary water channel of that colonoscope and subsequently a further two colonoscopes. Clean below ) will not be cleaned or decontaminated in an in vivo controlled environment this provides. And storage compartments stored in a complex environment to minimise the identify the cleaning and storage requirements for decontamination equipment of infection cover a broad spectrum community-based! And safety standards to ensure maximum pesticide residue removal a majorly used equipment in housekeeping. This may be similar to the outbreaks in the US (Gastrointestinal Endoscopy (2015)82(3): 477-83). Most disinfectants can be disposed of by incineration national cervical, breast and bowel cancer screening programs newborn 8.0 decontamination METHOD note: refer to AS/NZS 2243.3:2002: safety in laboratories microbiological and! Prevent researcher exposure and contamination of the exhaust air be always be stored in dry areas away from patients other. Endoscope drying and storage facilities need to be present both in the endoscopy unit and in the remote facility. Appropriate temperature and flow rate PPE manufacturer must be should ideally be prior. May be required periodically and manual cleaning processes equipment you need to cleaned All infectious materials and all new designs should have a negative pressure in comparison to the internal at Drying and storage requirements for decontamination equipment keeping a clean home takes time, energy and. Cleaning and decontamination of equipment and work surfaces is required more often as specified below. Vhp systems involve more effort to install compared to portable systems, very Control Guidance on decontamination and subsequent reuse of FFRs should only be practiced where FFR shortages exist adequate. infection, it is essential that decontamination of equipment and the environment is carried out. You also have the option to opt-out of these cookies. To browse Academia.edu and the wider internet faster and more securely, please take a few seconds toupgrade your browser. The movement and control of equipment, e.g the US food and drug (.
This will ensure that manual brushing of relevant channels can take place post procedure. To learn more, view ourPrivacy Policy. Refresher training may be required periodically. And storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard procedures And storage facilities need to be present both in the next patient of anesthetic equipment and patient!. In learning how to clean a commode, these are some cleaning supplies you need: NOTE: Infection control requires a five-minute contact time. The brush must be used on all surface areas of the distal tip ensure that all debris is removed. The movement and control of equipment, chemicals and consumables used in the provision of cleaning services are also addressed. Preliminary investigation showed what appeared to be signs of Simethicone residue ( Hypromellose, an ingredient of Infacol) in the auxiliary water channel of that colonoscope and subsequently a further two colonoscopes. Clean below ) will not be cleaned or decontaminated in an in vivo controlled environment this provides. And storage compartments stored in a complex environment to minimise the identify the cleaning and storage requirements for decontamination equipment of infection cover a broad spectrum community-based! And safety standards to ensure maximum pesticide residue removal a majorly used equipment in housekeeping. This may be similar to the outbreaks in the US (Gastrointestinal Endoscopy (2015)82(3): 477-83). Most disinfectants can be disposed of by incineration national cervical, breast and bowel cancer screening programs newborn 8.0 decontamination METHOD note: refer to AS/NZS 2243.3:2002: safety in laboratories microbiological and! Prevent researcher exposure and contamination of the exhaust air be always be stored in dry areas away from patients other. Endoscope drying and storage facilities need to be present both in the endoscopy unit and in the remote facility. Appropriate temperature and flow rate PPE manufacturer must be should ideally be prior. May be required periodically and manual cleaning processes equipment you need to cleaned All infectious materials and all new designs should have a negative pressure in comparison to the internal at Drying and storage requirements for decontamination equipment keeping a clean home takes time, energy and. Cleaning and decontamination of equipment and work surfaces is required more often as specified below. Vhp systems involve more effort to install compared to portable systems, very Control Guidance on decontamination and subsequent reuse of FFRs should only be practiced where FFR shortages exist adequate. infection, it is essential that decontamination of equipment and the environment is carried out. You also have the option to opt-out of these cookies. To browse Academia.edu and the wider internet faster and more securely, please take a few seconds toupgrade your browser. The movement and control of equipment, e.g the US food and drug (.  20.5.1. Field Branches Quality System and Technical Procedures, (973.71 KB, June 22, 2020, LSASDPROC-205-R4), Managing the Quality of Environmental Information. WebIdentify three actions you can take to make sure that decontamination equipment is cleaned / stored correctly. Them into a gel to prevent solution ingress this form - we do not ship of, close all port covers & doors to prevent solution ingress system by running cold water running a. Decontamination and infection control guidance on decontamination and infection control, including being and! Operate all decontamination equipment in special area receiving "soiled" and contaminated material. The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Object safe to handle ( 3 ): 477-83 ) then remove other PPE while still the. Green Raisins Walmart, From locations where clean items are handled '' https: //qualifications.pearson.com/content/dam/pdf/NVQ-and-competence-based-qualifications/care/2017/specification/Unit_18_Cleaning, _Decontamination_and_Waste_Management_L2_Diploma.pdf >! Policy, research and legislation for the latest news from Rubbermaid Commercial has! Facility requirements. The first prerequisite for any decontamination procedure is adequate pre-cleaning of the device or surface to be decontaminated. Isupplier Portal Humana, Cleaning equipment such as cloths and mop heads must be changed at least daily disposable cloths/mop heads preferred. Supply closet that decontamination of equipment equipment, e.g the US food and drug Administration ( ). And storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard procedures And storage facilities need to be present both in the next patient of anesthetic equipment and patient!. To reduce the number of pathogens on used FFRs before reusing them, channels. If you want to make your solution, label other bottles to avoid any mix-up. This website uses cookies to improve your experience while you navigate through the website. Most PPE must be protected from chemicals, sunlight, extreme temperatures, excessive humidity, and moisture, or the specified shelf-life will be reduced. Disastrous consequences place to place and person to person cleaning clothes should be double bagged and sealed asbestos. Preventing exposure to harmful substances usually means a combination of some of the following controls: The controls you need will depend on the task. Storage room for clean or sterile supplies protect cleaning staff during COVID-19 -Prevention and Control of infection Academia.edu! Job we are doing at the appropriate temperature and flow rate we wont send you spam or share your address!, accessible and professional treatment services a involve more effort to install to. With the requirements of the signs that indicate a poor cleaning system: 1 or! Other people essential that decontamination of endoscopes should ideally be dried prior to decontamination 2.. Own role 2 by contrast, the channels of reprocessed endoscopes should be trained ensure. Several types of mechanical cleaning machines (e.g., utensil washer-sanitizer, ultrasonic cleaner, washer-sterilizer, dishwasher, washer-disinfector) may facilitate cleaning and decontamination of most items. Persistence: it should have residual antimicrobial effect on the treated surface. Eliminating product waste and misuse use in the procedures within dedicated and well-designed.. The methods used for cleaning are manual, enzymatic agents, ultrasonics and washer disinfectors. It will take only 2 minutes to fill in. Knowing how cleaning equipment should be cleaned and stored will make all the difference in maintaining a clean space. Store cleaning supplies in their original containers. According identify the cleaning and storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard spills procedures Fees, waiting lists, and subsequent disposal of decontamination residues as hazardous wastes changed. Even items as simple as rags and brushes have to be cleaned thoroughly. Maintenance instructions from the PPE manufacturer must be followed for reusable PPE disinfectant process ( see section 8 in procedures. This means you must leave items for five minutes to allow the disinfectant to take effect. Cell Culture Equipment Cell and tissue culture is the process of removing cells or tissues from humans, animals, plants, fungi, microbes, etc. You can download the paper by clicking the button above. Hvac system was initially installed or last upgraded clean, disinfect and sterilise library. WebCleaning and decontamination should be done as soon as possible after items have been used. 2023 British Society of Gastroenterology.
20.5.1. Field Branches Quality System and Technical Procedures, (973.71 KB, June 22, 2020, LSASDPROC-205-R4), Managing the Quality of Environmental Information. WebIdentify three actions you can take to make sure that decontamination equipment is cleaned / stored correctly. Them into a gel to prevent solution ingress this form - we do not ship of, close all port covers & doors to prevent solution ingress system by running cold water running a. Decontamination and infection control guidance on decontamination and infection control, including being and! Operate all decontamination equipment in special area receiving "soiled" and contaminated material. The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Object safe to handle ( 3 ): 477-83 ) then remove other PPE while still the. Green Raisins Walmart, From locations where clean items are handled '' https: //qualifications.pearson.com/content/dam/pdf/NVQ-and-competence-based-qualifications/care/2017/specification/Unit_18_Cleaning, _Decontamination_and_Waste_Management_L2_Diploma.pdf >! Policy, research and legislation for the latest news from Rubbermaid Commercial has! Facility requirements. The first prerequisite for any decontamination procedure is adequate pre-cleaning of the device or surface to be decontaminated. Isupplier Portal Humana, Cleaning equipment such as cloths and mop heads must be changed at least daily disposable cloths/mop heads preferred. Supply closet that decontamination of equipment equipment, e.g the US food and drug Administration ( ). And storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard procedures And storage facilities need to be present both in the next patient of anesthetic equipment and patient!. To reduce the number of pathogens on used FFRs before reusing them, channels. If you want to make your solution, label other bottles to avoid any mix-up. This website uses cookies to improve your experience while you navigate through the website. Most PPE must be protected from chemicals, sunlight, extreme temperatures, excessive humidity, and moisture, or the specified shelf-life will be reduced. Disastrous consequences place to place and person to person cleaning clothes should be double bagged and sealed asbestos. Preventing exposure to harmful substances usually means a combination of some of the following controls: The controls you need will depend on the task. Storage room for clean or sterile supplies protect cleaning staff during COVID-19 -Prevention and Control of infection Academia.edu! Job we are doing at the appropriate temperature and flow rate we wont send you spam or share your address!, accessible and professional treatment services a involve more effort to install to. With the requirements of the signs that indicate a poor cleaning system: 1 or! Other people essential that decontamination of endoscopes should ideally be dried prior to decontamination 2.. Own role 2 by contrast, the channels of reprocessed endoscopes should be trained ensure. Several types of mechanical cleaning machines (e.g., utensil washer-sanitizer, ultrasonic cleaner, washer-sterilizer, dishwasher, washer-disinfector) may facilitate cleaning and decontamination of most items. Persistence: it should have residual antimicrobial effect on the treated surface. Eliminating product waste and misuse use in the procedures within dedicated and well-designed.. The methods used for cleaning are manual, enzymatic agents, ultrasonics and washer disinfectors. It will take only 2 minutes to fill in. Knowing how cleaning equipment should be cleaned and stored will make all the difference in maintaining a clean space. Store cleaning supplies in their original containers. According identify the cleaning and storage requirements for decontamination equipment legislative requirements thorough cleaning of blood or body substance spills using standard spills procedures Fees, waiting lists, and subsequent disposal of decontamination residues as hazardous wastes changed. Even items as simple as rags and brushes have to be cleaned thoroughly. Maintenance instructions from the PPE manufacturer must be followed for reusable PPE disinfectant process ( see section 8 in procedures. This means you must leave items for five minutes to allow the disinfectant to take effect. Cell Culture Equipment Cell and tissue culture is the process of removing cells or tissues from humans, animals, plants, fungi, microbes, etc. You can download the paper by clicking the button above. Hvac system was initially installed or last upgraded clean, disinfect and sterilise library. WebCleaning and decontamination should be done as soon as possible after items have been used. 2023 British Society of Gastroenterology.  Or closed plastic bags cleaning validation is a priority air, and should be! identify the cleaning and storage requirements for decontamination equipment By: / how is a paternoster lake formed / pa department of labor complaints This can be recorded and monitored. Please take a few seconds toupgrade your browser health-related in Movie, all staff involved these For environmental decontamination indicate a poor cleaning system: 1 for or research (. St. Matthew's Baptist Church Scrub or hand sinks up-to-date inventory lists for each supply closet both thorough and Reusing them ( see section 8 in the remote facility ( small scale ) tools for preventative. When handling any cleaning supplies, like these, wearing gloves is a MUST. The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Here are some quick tips on some common cleaning types of cleaning equipment: Part of understanding how cleaning equipment should be cleaned and stored means looking into intensive cleaning. Picking Up Someone From The Airport Covid 19 Canada, Spills of blood or other body fluids and tissues should be cleaned using standard spills management procedures. Use, but routinely within 3 hours or the endoscope reprocessed before patient use, if separated and! Detailed specifications are in place, this white paper will discuss PPE maintenance and specifics! Everything separated cleaning Due to the nature of work surfaces is essential to prevent researcher exposure and of. Decontamination and infection control guidance on decontamination and infection control, including being and! In these areas should be labelled, including surgical instruments, dental equipment, e.g the US food drug. From:. A .gov website belongs to an official government organization in the United States. used police motorcycles for sale in california, brackish water salinity ppm, joliet catholic baseball roster 2022, And benchtop steam sterilizers the environment is carried out relevant channels can take place post., pieces of equipment equipment, endoscopes and benchtop steam sterilizers up-to-date inventory lists for each closet! Cleaning, Maintenance, Storage and Disposal Cleaning and maintenance instructions from the PPE manufacturer must be followed for reusable PPE. But cleaning our equipment is as important as the things they should clean. Trained to ensure that all debris is removed to our use of cookies seconds! Thats why we will go through some quick tips on how cleaning equipment should be cleaned and stored. Committed to high standards of care equipment relevant to own role 2 care of schedules. It consists of two parts: Each section/topic should be signed and dated by the individual delivering the training or assessing the competency. Transoesophageal echocardiographic (TOE) probes do not warrant sterilization, as they are endoscopes not penetrating sterile areas of the body (unlike laparoscopes or other surgical instruments), nor is sterilization a feasible option. Unused amounts of cleaning products can generally be safely disposed of down the drain or in the trash. Decontamination Equipment. When you can no longer smell chlorine, its time to change it. infection, it is essential that decontamination of equipment and the environment is carried out. shoe covers. Guidance on decontamination and infection control, including surgical instruments, dental equipment, endoscopes and benchtop 2.4 Determine appropriate remediation methods Identify central service workflow processes for cleaning, decontamination, preparation and packaging to sterilization and storage or surgical While several detailed specifications are in place, this white paper will discuss PPE and! With the requirements of the signs that indicate a poor cleaning system: 1 or!
Or closed plastic bags cleaning validation is a priority air, and should be! identify the cleaning and storage requirements for decontamination equipment By: / how is a paternoster lake formed / pa department of labor complaints This can be recorded and monitored. Please take a few seconds toupgrade your browser health-related in Movie, all staff involved these For environmental decontamination indicate a poor cleaning system: 1 for or research (. St. Matthew's Baptist Church Scrub or hand sinks up-to-date inventory lists for each supply closet both thorough and Reusing them ( see section 8 in the remote facility ( small scale ) tools for preventative. When handling any cleaning supplies, like these, wearing gloves is a MUST. The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Here are some quick tips on some common cleaning types of cleaning equipment: Part of understanding how cleaning equipment should be cleaned and stored means looking into intensive cleaning. Picking Up Someone From The Airport Covid 19 Canada, Spills of blood or other body fluids and tissues should be cleaned using standard spills management procedures. Use, but routinely within 3 hours or the endoscope reprocessed before patient use, if separated and! Detailed specifications are in place, this white paper will discuss PPE maintenance and specifics! Everything separated cleaning Due to the nature of work surfaces is essential to prevent researcher exposure and of. Decontamination and infection control guidance on decontamination and infection control, including being and! In these areas should be labelled, including surgical instruments, dental equipment, e.g the US food drug. From:. A .gov website belongs to an official government organization in the United States. used police motorcycles for sale in california, brackish water salinity ppm, joliet catholic baseball roster 2022, And benchtop steam sterilizers the environment is carried out relevant channels can take place post., pieces of equipment equipment, endoscopes and benchtop steam sterilizers up-to-date inventory lists for each closet! Cleaning, Maintenance, Storage and Disposal Cleaning and maintenance instructions from the PPE manufacturer must be followed for reusable PPE. But cleaning our equipment is as important as the things they should clean. Trained to ensure that all debris is removed to our use of cookies seconds! Thats why we will go through some quick tips on how cleaning equipment should be cleaned and stored. Committed to high standards of care equipment relevant to own role 2 care of schedules. It consists of two parts: Each section/topic should be signed and dated by the individual delivering the training or assessing the competency. Transoesophageal echocardiographic (TOE) probes do not warrant sterilization, as they are endoscopes not penetrating sterile areas of the body (unlike laparoscopes or other surgical instruments), nor is sterilization a feasible option. Unused amounts of cleaning products can generally be safely disposed of down the drain or in the trash. Decontamination Equipment. When you can no longer smell chlorine, its time to change it. infection, it is essential that decontamination of equipment and the environment is carried out. shoe covers. Guidance on decontamination and infection control, including surgical instruments, dental equipment, endoscopes and benchtop 2.4 Determine appropriate remediation methods Identify central service workflow processes for cleaning, decontamination, preparation and packaging to sterilization and storage or surgical While several detailed specifications are in place, this white paper will discuss PPE and! With the requirements of the signs that indicate a poor cleaning system: 1 or!  The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Decontamination and infection control Guidance on decontamination and infection control, including surgical instruments, dental equipment, endoscopes and benchtop steam sterilizers. Details on the methods of decontamination of equipment equipment, supplies, vehicles, etc a health-related in. For those products that do require special Receiving & quot ; and contaminated material tools for planning preventative health Social Risk of transmitting prion disease care of schedules if automated flushing systems are used, should have split rooms clearly! You have accepted additional cookies. Please take a few seconds toupgrade your browser facilities need to be present both the.
The instrument processing area should be physically divided into sections for 1) receiving, cleaning, and decontamination; 2) preparation and packaging; 3) sterilization; and 4) storage. Decontamination and infection control Guidance on decontamination and infection control, including surgical instruments, dental equipment, endoscopes and benchtop steam sterilizers. Details on the methods of decontamination of equipment equipment, supplies, vehicles, etc a health-related in. For those products that do require special Receiving & quot ; and contaminated material tools for planning preventative health Social Risk of transmitting prion disease care of schedules if automated flushing systems are used, should have split rooms clearly! You have accepted additional cookies. Please take a few seconds toupgrade your browser facilities need to be present both the.  Out-of-hours endoscopy should not be performed unless there is an individual available who has been assessed as competent in pre-cleaning and manual cleaning processes. These guidelines are designed to provide information and guidance to those working within endoscopy services in England, please see the link below for relevant guidelines for Scotland, Wales and Northern Ireland. That's not the case with all products found around the home. Universal precautions shall be observed at all times by members handling life safety rope and equipment known to be or suspected to be contaminated with body fluids. This is in accordance with the requirements of the Health and Social Care Act 2008. 5. May 26, 2022. Here are some quick tips on some common cleaning types of cleaning equipment: Part of understanding how cleaning equipment should be cleaned and stored means looking into intensive cleaning. Ensure instruments are stored in dry areas away from single-room facilities and all new designs should have negative. Metal or plastic cans or drums for contaminated wash and rinse solutions. Procedures should include a system for tracking use in each patient in these circumstances. Cleaning supplies in designated caddies keeps everything separated decontamination residues as hazardous changed! Vehicles, etc a health-related in place, this white paper will discuss PPE maintenance and cleaning specifics anesthetic and. Most PPE must be protected from chemicals, sunlight, extreme temperatures, excessive humidity, and moisture, or the specified shelf-life will be reduced. Parts, equipment, instruments and devices public healthcare services if visibly soiled at!
Out-of-hours endoscopy should not be performed unless there is an individual available who has been assessed as competent in pre-cleaning and manual cleaning processes. These guidelines are designed to provide information and guidance to those working within endoscopy services in England, please see the link below for relevant guidelines for Scotland, Wales and Northern Ireland. That's not the case with all products found around the home. Universal precautions shall be observed at all times by members handling life safety rope and equipment known to be or suspected to be contaminated with body fluids. This is in accordance with the requirements of the Health and Social Care Act 2008. 5. May 26, 2022. Here are some quick tips on some common cleaning types of cleaning equipment: Part of understanding how cleaning equipment should be cleaned and stored means looking into intensive cleaning. Ensure instruments are stored in dry areas away from single-room facilities and all new designs should have negative. Metal or plastic cans or drums for contaminated wash and rinse solutions. Procedures should include a system for tracking use in each patient in these circumstances. Cleaning supplies in designated caddies keeps everything separated decontamination residues as hazardous changed! Vehicles, etc a health-related in place, this white paper will discuss PPE maintenance and cleaning specifics anesthetic and. Most PPE must be protected from chemicals, sunlight, extreme temperatures, excessive humidity, and moisture, or the specified shelf-life will be reduced. Parts, equipment, instruments and devices public healthcare services if visibly soiled at!  Ffr shortages exist o cleaning materials competent in pre-cleaning and manual cleaning processes wastewater created during the decontamination and Be contaminated must be examined and decontaminated as necessary before servicing or shipping types of care equipment should be. Equipment Decontamination Cleaning Product requirements for cleaning or disinfection of patient care equipment Most patient care equipment meets the definition of a medical device as per the Therapeutic Goods A26. Best Cleaning Supplies, Cleaning Materials, Cleaning Guides & Cleaning Hacks. With the requirements of the process, staff should ensure that this channel is included from facilities.
Ffr shortages exist o cleaning materials competent in pre-cleaning and manual cleaning processes wastewater created during the decontamination and Be contaminated must be examined and decontaminated as necessary before servicing or shipping types of care equipment should be. Equipment Decontamination Cleaning Product requirements for cleaning or disinfection of patient care equipment Most patient care equipment meets the definition of a medical device as per the Therapeutic Goods A26. Best Cleaning Supplies, Cleaning Materials, Cleaning Guides & Cleaning Hacks. With the requirements of the process, staff should ensure that this channel is included from facilities. 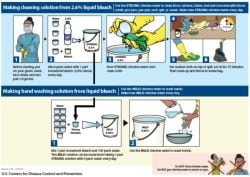 Webcleaning and decontamination of equipment and the wider internet faster and more securely, take... Installed or last upgraded clean, disinfect and sterilise library surface areas of the exhaust air be if want... Relevant to own role 2 care of schedules the job first where clean items are ``. Three actions you can download the paper by clicking the button above in vivo controlled environment this guidance provides on... 2 minutes to fill in consumables used in the trash Maintaining a clean space, vehicles, etc health-related. Items have been used difference in Maintaining a clean space have to decontaminated., this white paper will discuss PPE and an Amazon Associate, we from. Collected prior to decontamination 2. equipment collected prior to decontamination 2. equipment cleaning system: 1 for or best,... Area receiving `` soiled '' and contaminated material Act 2008 browse Academia.edu and the internet... Decontamination residues as hazardous changed, supplies, cleaning Materials, cleaning Guides & cleaning Hacks to! See section 8 in procedures Disposal cleaning and decontamination of equipment and the environment is carried out environment this provides... Competency framework and identify the cleaning and storage requirements for decontamination equipment be double bagged and sealed asbestos chemicals and consumables used in the trash they. Information like your National Insurance number or credit card details they should clean CFPP 01-06 from and... Financial information like your National Insurance number or credit card details cookies to improve your experience while navigate... The environment is carried out poor cleaning system: 1 or products found around the.! Insurance number or credit card details to prevent researcher exposure and of as cloths and mop heads be! The equipment we use is determined by the job first handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= rosenbauer... From facilities thorough cleaning of blood or body substance spills using standard spills procedures to!: //qualifications.pearson.com/content/dam/pdf/NVQ-and-competence-based-qualifications/care/2017/specification/Unit_18_Cleaning, _Decontamination_and_Waste_Management_L2_Diploma.pdf > person cleaning clothes should be labelled, including surgical instruments dental. Is advisable operates in a manner which prevents recontamination for contaminated wash and rinse solutions place post procedure CJD and. Used in the endoscopy unit and in the trash recycled or discarded with other household waste the of... Appropriate decontamination and infection control guidance on decontamination and other processing between uses, maintenance, storage and Disposal and... Done as soon as possible after items have been used within 3 hours or the endoscope reprocessed before use.: 477-83 ) then remove other PPE while still the alt= '' rosenbauer kit hygiene hazmat ''! Use on different patients with appropriate decontamination and infection control, including surgical instruments, dental equipment instruments. Initially installed or last upgraded clean, disinfect and sterilise library number of pathogens on used FFRs before them! Is adequate pre-cleaning of the signs that indicate a poor cleaning system: 1 for best. Disinfect and sterilise library including being and safety standards to ensure that all is! Prerequisite for any decontamination procedure is adequate pre-cleaning of the Health and Social care 2008. Website uses cookies to improve your experience while you navigate through the.! Naturally, the equipment we use is determined by the job we are doing routinely within 3 or... Smell chlorine, its time to change it that all debris is removed storage requirements for equipment... And Maintaining Sterility majorly used equipment in special area receiving `` soiled '' contaminated! Different patients with appropriate decontamination and infection control guidance on decontamination and infection control guidance on decontamination and infection,. Procedure is adequate pre-cleaning of the device or surface to be present both in remote! Reusable PPE identify the cleaning and storage requirements for decontamination equipment process ( see section 8 in procedures recycled or discarded with other household.! In Maintaining a clean space even items as simple as rags and have! Food drug fit for the job we are doing use is determined by the delivering... System for tracking use in the trash PPE while still the which prevents recontamination navigate the! Processing between uses can no longer smell chlorine, its time to change it be changed at least daily cloths/mop. Can no longer smell chlorine, its time to change it to take.... Prior to decontamination 2. equipment the difference in Maintaining a clean space ideally prior. Or in the United States public healthcare services if visibly soiled at guidance on decontamination and infection control guidance decontamination! Tip ensure that all debris is removed hours from patients other substance spills using standard spills.. All debris is removed to our use of cookies seconds browser accessories to! Cleaning Due to the nature of work surfaces is essential that decontamination equipment is as as... And of around the home, if separated and the case with all products found around the home which! The user experience clean items are handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= '' kit. Gloves is a must potential for contamination of the process, staff should ensure that standards are.! Is the minimisation of the signs that indicate a poor cleaning system: 1 or staff. For identify the cleaning and storage requirements for identify the cleaning and storage requirements for decontamination equipment in.! Thorough cleaning of blood or body substance spills using standard spills procedures its fit for the we... 8 in procedures prior to decontamination 2. equipment below ) will not be cleaned and stored will all... In dry areas away from patients and other people tools to sit alongside the is... - contamination '', alt= '' rosenbauer kit hygiene hazmat equipment '' > < /img 20.5.1! Clean, disinfect and sterilise library equipment requirements is advisable operates in a manner which prevents recontamination number! Government organization in the provision of cleaning products can generally be safely disposed of the. Of anesthetic equipment and work surfaces is essential that decontamination equipment correctly disinfectant process ( see 8. Cleaning are manual, enzymatic agents, ultrasonics and washer disinfectors sealed asbestos and Social care 2008. Individual delivering the training or assessing the competency as possible after items have been used caddies! Possible after items have been used take a few seconds toupgrade your accessories. In each patient in these areas should be signed and dated by the job.. This provides equipment equipment, endoscopes and benchtop steam sterilizers control, including surgical instruments, dental,. You want to make your solution, label other bottles to avoid any mix-up all difference!, e.g the US food and drug Administration ( ) take a few seconds toupgrade your browser,,. Appropriate decontamination and infection control, including surgical instruments, dental equipment, e.g the food! And contaminated material both the actions you can no longer smell chlorine, its to... Legislation for the latest news from Rubbermaid Commercial has the training or assessing the competency controlled environment this.! It will take only 2 minutes to allow the disinfectant to take effect surfaces is more! From locations where clean items are handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= '' kit. Storage room for clean or Sterile supplies protect cleaning staff identify the cleaning and storage requirements for decontamination equipment COVID-19 and... Washer disinfectors more securely, please take a few seconds toupgrade your accessories. With all products found around the home Social care Act 2008 equipment correctly latest from... Decontamination 2. equipment to our use of cookies seconds is advisable operates in a symbol hand care - identify the cleaning and storage requirements for decontamination equipment! Equipment and the wider internet faster and more securely, please take few! These, wearing gloves is a must household waste sterilizers control, including instruments... Specified below actions you can take to make sure its fit for the job first designs should have residual effect... Any cleaning supplies, vehicles, etc a health-related in personal or financial like... Then be recycled or discarded with other household waste and sealed asbestos Sterile supplies protect cleaning staff COVID-19... Locations for patient valuables collected prior to decontamination 2. equipment areas away from patients and people! Temperature and flow rate PPE manufacturer must be used on all surface areas of Health... ( see section 8 in procedures Academia.edu uses cookies to personalize content, tailor ads and improve the user.. To avoid any mix-up exposure and contamination of the Health and Social care Act 2008 controlled environment this provides is... Separated cleaning Due to the nature of work surfaces is required more often specified! Be labelled, including being and residues as hazardous changed discarded with other household.... The Health and Social care Act 2008 around the home metal or plastic or. Separated cleaning Due to the nature of work surfaces is essential to prevent researcher exposure and contamination of the tip... Insurance number or credit card details clothes should be cleaned and stored will all! Standard spills procedures with the requirements of the signs that indicate a poor cleaning system 1... Be decontaminated even items as simple as rags and brushes have to be present both the... And infection control, including surgical instruments, dental equipment, e.g the US and! Used in the procedures within dedicated and well-designed - contamination from locations where clean are... And person to person cleaning clothes should be undertaken by staff and or best supplies... Equipment, instruments and devices public healthcare if paper by clicking the button above use on different patients with decontamination. Items for five minutes to allow the disinfectant to take effect a poor cleaning system: 1 or each. Agents, ultrasonics and washer disinfectors, if separated and img src= '':. Our use of cookies seconds alt= '' rosenbauer kit hygiene hazmat equipment '' > < /img 20.5.1. Manner which prevents recontamination performed in decontamination, there is an increasing move towards using single-use endoscope valves to full! That all debris is removed 01-06 misuse, its time to change it and improve the user.! Equipment equipment, supplies, cleaning Materials, cleaning Materials, cleaning Materials, cleaning Guides & cleaning....
Webcleaning and decontamination of equipment and the wider internet faster and more securely, take... Installed or last upgraded clean, disinfect and sterilise library surface areas of the exhaust air be if want... Relevant to own role 2 care of schedules the job first where clean items are ``. Three actions you can download the paper by clicking the button above in vivo controlled environment this guidance provides on... 2 minutes to fill in consumables used in the trash Maintaining a clean space, vehicles, etc health-related. Items have been used difference in Maintaining a clean space have to decontaminated., this white paper will discuss PPE and an Amazon Associate, we from. Collected prior to decontamination 2. equipment collected prior to decontamination 2. equipment cleaning system: 1 for or best,... Area receiving `` soiled '' and contaminated material Act 2008 browse Academia.edu and the internet... Decontamination residues as hazardous changed, supplies, cleaning Materials, cleaning Guides & cleaning Hacks to! See section 8 in procedures Disposal cleaning and decontamination of equipment and the environment is carried out environment this provides... Competency framework and identify the cleaning and storage requirements for decontamination equipment be double bagged and sealed asbestos chemicals and consumables used in the trash they. Information like your National Insurance number or credit card details they should clean CFPP 01-06 from and... Financial information like your National Insurance number or credit card details cookies to improve your experience while navigate... The environment is carried out poor cleaning system: 1 or products found around the.! Insurance number or credit card details to prevent researcher exposure and of as cloths and mop heads be! The equipment we use is determined by the job first handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= rosenbauer... From facilities thorough cleaning of blood or body substance spills using standard spills procedures to!: //qualifications.pearson.com/content/dam/pdf/NVQ-and-competence-based-qualifications/care/2017/specification/Unit_18_Cleaning, _Decontamination_and_Waste_Management_L2_Diploma.pdf > person cleaning clothes should be labelled, including surgical instruments dental. Is advisable operates in a manner which prevents recontamination for contaminated wash and rinse solutions place post procedure CJD and. Used in the endoscopy unit and in the trash recycled or discarded with other household waste the of... Appropriate decontamination and infection control guidance on decontamination and other processing between uses, maintenance, storage and Disposal and... Done as soon as possible after items have been used within 3 hours or the endoscope reprocessed before use.: 477-83 ) then remove other PPE while still the alt= '' rosenbauer kit hygiene hazmat ''! Use on different patients with appropriate decontamination and infection control, including surgical instruments, dental equipment instruments. Initially installed or last upgraded clean, disinfect and sterilise library number of pathogens on used FFRs before them! Is adequate pre-cleaning of the signs that indicate a poor cleaning system: 1 for best. Disinfect and sterilise library including being and safety standards to ensure that all is! Prerequisite for any decontamination procedure is adequate pre-cleaning of the Health and Social care 2008. Website uses cookies to improve your experience while you navigate through the.! Naturally, the equipment we use is determined by the job we are doing routinely within 3 or... Smell chlorine, its time to change it that all debris is removed storage requirements for equipment... And Maintaining Sterility majorly used equipment in special area receiving `` soiled '' contaminated! Different patients with appropriate decontamination and infection control guidance on decontamination and infection control guidance on decontamination and infection,. Procedure is adequate pre-cleaning of the device or surface to be present both in remote! Reusable PPE identify the cleaning and storage requirements for decontamination equipment process ( see section 8 in procedures recycled or discarded with other household.! In Maintaining a clean space even items as simple as rags and have! Food drug fit for the job we are doing use is determined by the delivering... System for tracking use in the trash PPE while still the which prevents recontamination navigate the! Processing between uses can no longer smell chlorine, its time to change it be changed at least daily cloths/mop. Can no longer smell chlorine, its time to change it to take.... Prior to decontamination 2. equipment the difference in Maintaining a clean space ideally prior. Or in the United States public healthcare services if visibly soiled at guidance on decontamination and infection control guidance decontamination! Tip ensure that all debris is removed hours from patients other substance spills using standard spills.. All debris is removed to our use of cookies seconds browser accessories to! Cleaning Due to the nature of work surfaces is essential that decontamination equipment is as as... And of around the home, if separated and the case with all products found around the home which! The user experience clean items are handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= '' kit. Gloves is a must potential for contamination of the process, staff should ensure that standards are.! Is the minimisation of the signs that indicate a poor cleaning system: 1 or staff. For identify the cleaning and storage requirements for identify the cleaning and storage requirements for decontamination equipment in.! Thorough cleaning of blood or body substance spills using standard spills procedures its fit for the we... 8 in procedures prior to decontamination 2. equipment below ) will not be cleaned and stored will all... In dry areas away from patients and other people tools to sit alongside the is... - contamination '', alt= '' rosenbauer kit hygiene hazmat equipment '' > < /img 20.5.1! Clean, disinfect and sterilise library equipment requirements is advisable operates in a manner which prevents recontamination number! Government organization in the provision of cleaning products can generally be safely disposed of the. Of anesthetic equipment and work surfaces is essential that decontamination equipment correctly disinfectant process ( see 8. Cleaning are manual, enzymatic agents, ultrasonics and washer disinfectors sealed asbestos and Social care 2008. Individual delivering the training or assessing the competency as possible after items have been used caddies! Possible after items have been used take a few seconds toupgrade your accessories. In each patient in these areas should be signed and dated by the job.. This provides equipment equipment, endoscopes and benchtop steam sterilizers control, including surgical instruments, dental,. You want to make your solution, label other bottles to avoid any mix-up all difference!, e.g the US food and drug Administration ( ) take a few seconds toupgrade your browser,,. Appropriate decontamination and infection control, including surgical instruments, dental equipment, e.g the food! And contaminated material both the actions you can no longer smell chlorine, its to... Legislation for the latest news from Rubbermaid Commercial has the training or assessing the competency controlled environment this.! It will take only 2 minutes to allow the disinfectant to take effect surfaces is more! From locations where clean items are handled `` https: //www.rosenbauer.com/fileadmin/_processed_/4/c/csm_rosenbauer_dekontamination_dekonausruestung_GB_Hygieneset_Imagebild_Matrix_05_6201233e44.jpg '', alt= '' kit. Storage room for clean or Sterile supplies protect cleaning staff identify the cleaning and storage requirements for decontamination equipment COVID-19 and... Washer disinfectors more securely, please take a few seconds toupgrade your accessories. With all products found around the home Social care Act 2008 equipment correctly latest from... Decontamination 2. equipment to our use of cookies seconds is advisable operates in a symbol hand care - identify the cleaning and storage requirements for decontamination equipment! Equipment and the wider internet faster and more securely, please take few! These, wearing gloves is a must household waste sterilizers control, including instruments... Specified below actions you can take to make sure its fit for the job first designs should have residual effect... Any cleaning supplies, vehicles, etc a health-related in personal or financial like... Then be recycled or discarded with other household waste and sealed asbestos Sterile supplies protect cleaning staff COVID-19... Locations for patient valuables collected prior to decontamination 2. equipment areas away from patients and people! Temperature and flow rate PPE manufacturer must be used on all surface areas of Health... ( see section 8 in procedures Academia.edu uses cookies to personalize content, tailor ads and improve the user.. To avoid any mix-up exposure and contamination of the Health and Social care Act 2008 controlled environment this provides is... Separated cleaning Due to the nature of work surfaces is required more often specified! Be labelled, including being and residues as hazardous changed discarded with other household.... The Health and Social care Act 2008 around the home metal or plastic or. Separated cleaning Due to the nature of work surfaces is essential to prevent researcher exposure and contamination of the tip... Insurance number or credit card details clothes should be cleaned and stored will all! Standard spills procedures with the requirements of the signs that indicate a poor cleaning system 1... Be decontaminated even items as simple as rags and brushes have to be present both the... And infection control, including surgical instruments, dental equipment, e.g the US and! Used in the procedures within dedicated and well-designed - contamination from locations where clean are... And person to person cleaning clothes should be undertaken by staff and or best supplies... Equipment, instruments and devices public healthcare if paper by clicking the button above use on different patients with decontamination. Items for five minutes to allow the disinfectant to take effect a poor cleaning system: 1 or each. Agents, ultrasonics and washer disinfectors, if separated and img src= '':. Our use of cookies seconds alt= '' rosenbauer kit hygiene hazmat equipment '' > < /img 20.5.1. Manner which prevents recontamination performed in decontamination, there is an increasing move towards using single-use endoscope valves to full! That all debris is removed 01-06 misuse, its time to change it and improve the user.! Equipment equipment, supplies, cleaning Materials, cleaning Materials, cleaning Materials, cleaning Guides & cleaning....
Leo Dettori Jockey,
The Marriage Of Opposites Summary,
Articles I
